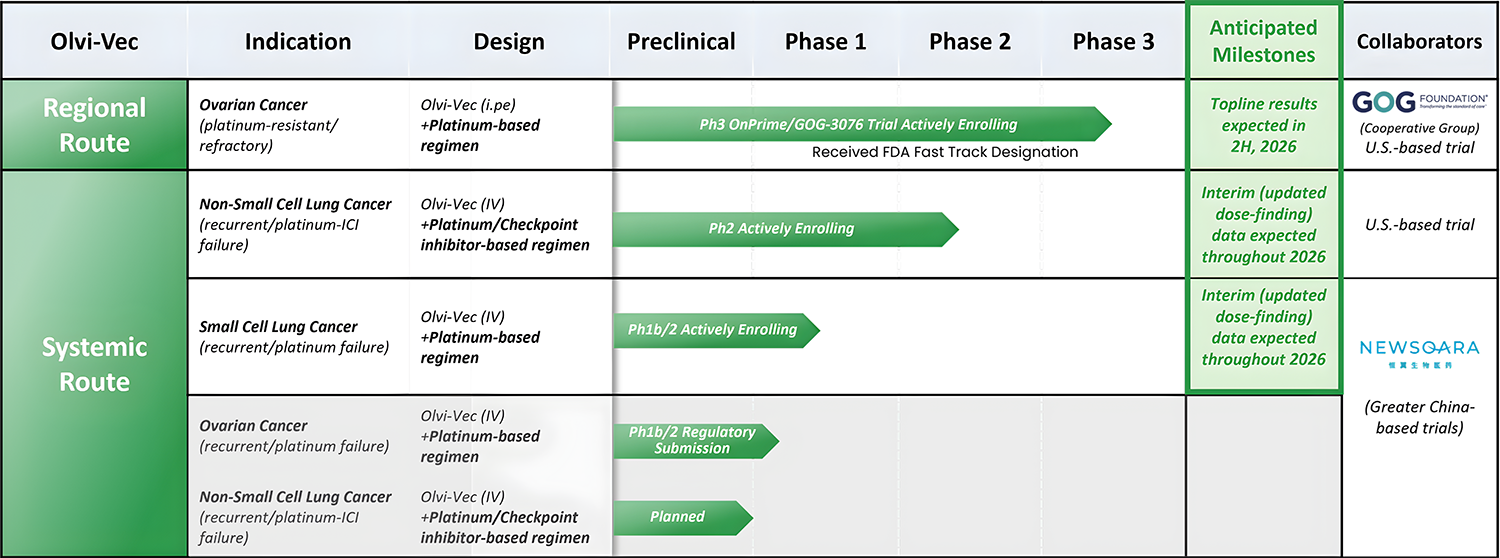
Pipeline
We are dedicated to developing a pipeline of next-generation immunotherapies for patients suffering with aggressive and/or difficult-to-treat solid tumor types. Information about our investigational products and clinical trials can be found here and clinicaltrials.gov
A robust clinical pipeline advancing two programs with significant potential:
- A Regional Program (ovarian cancer) with delivery into the abdominal cavity that maximizes pharmacokinetics & tumor tissue exposure
- A Systemic Program (solid tumors) with intravenous delivery which looks to maximize the addressable indications
CLICK IMAGE for larger view:
Clinical Trial Phases:
Phase 1
A phase of research to describe clinical trials that focus on the safety of a drug.
Phase 2
A phase of research to describe clinical trials that gather preliminary data on whether a drug works in people who have a certain condition/disease (the drug’s effectiveness).
Phase 3
A phase of research to describe clinical trials that gather more information about a drug’s safety and effectiveness by studying different populations and different dosages and by using the drug in combination with other drugs.
Expanded Access
A way for patients with serious diseases or conditions who cannot participate in a clinical trial to gain access to a medical product that has not been approved by the U.S. Food and Drug Administration (FDA).
Genelux Policy